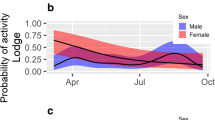
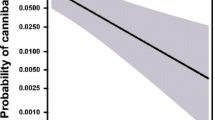
Abstract
The immune response can be costly. Studies in several arthropod species have indicated a trade-off between immunity and other life-history traits, including reproduction. In sexually dimorphic species in which females and males largely differ in their life history strategies and related energetic demands, we can expect to find sex differences in immune functions. Sex differences in immunity are well documented in vertebrates; however, we largely lack data from invertebrate systems. Lytic activity, the immune system’s ability to lysate bacteria and viruses, has been widely used as a proxy for the strength of the immune response in several invertebrates. With this in mind, we used the burrowing wolf spider Allocosa senex to test differences in lytic activity between females and males. We also studied whether digging behavior affects the immune responses in this species. While females of A. senex construct simple refuges where they stay during the day, males construct deep burrows, which they donate to females after copulation. In accordance with our hypothesis, females showed higher lytic activity compared with males, and those males who dug showed higher levels of lytic activity than those that did not dig. Furthermore, male body condition and lytic activity did not correlate with burrow length, a trait under female choice in this species. Our results show sexual dimorphism in lytic activity responses, which are likely related to differences in life-history strategies and energetic requirements of each sex in A. senex spiders.

Similar content being viewed by others
References
Adamo SA, Spiteri RJ (2005) Female choice for male immunocompetence: when is it worth it? Behav Ecol 16:871–879. https://doi.org/10.1093/beheco/ari068
Adamo SA, Spiteri RJ (2009) He’s healthy, but will he survive the plague? Possible constraints on mate choice for disease resistance. Anim Behav 77:67–78. https://doi.org/10.1016/j.anbehav.2008.09.011
Ahmed AM, Hurd H (2006) Immune stimulation and malaria infection impose reproductive costs in Anopheles gambiae via follicular apoptosis. Microbes Infect 8:308–315. https://doi.org/10.1016/j.micinf.2005.06.026
Ahtiainen JJ, Alatalo RV, Kortet R, Rantala MJ (2004) Sexual advertisement and immune function in an arachnid species (Lycosidae). Behav Ecol 15:602–606. https://doi.org/10.1093/beheco/arh062
Ahtiainen JJ, Alatalo RV, Kortet R, Rantala MJ (2005) A tradeoff between sexual signalling and immune function in a natural population of the drumming wolf spider Hygrolycosa rubrofasciata. J Evol Biol 18:985–991. https://doi.org/10.1111/j.1420-9101.2005.00907.x
Ahtiainen JJ, Alatalo RV, Kortet R, Rantala MJ (2006) Immune function, dominance and mating success in drumming male wolf spiders Hygrolycosa rubrofasciata. Behav Ecol Sociobiol 60:826–832. https://doi.org/10.1007/s00265-006-0226-9
Aisenberg A, Peretti AV (2011a) Sexual dimorphism in immune response, fat reserves and muscle mass in a sex role reversed spider. Zoology 114:272–275. https://doi.org/10.1016/j.zool.2011.05.003
Aisenberg A, Peretti AV (2011b) Male burrow digging in a sex role-reversed spider inhabiting water-margin environment. Bull Br Arachnol Soc 15:201–204. https://doi.org/10.13156/arac.2011.15.6.201
Aisenberg A, Viera C, Costa FG (2007) Daring females, devoted males, and reversed sexual size dimorphism in the sand-dwelling spider Allocosa brasiliensis (Araneae, Lycosidae). Behav Ecol Sociobiol 62:29–35. https://doi.org/10.1007/s00265-007-0435-x
Andersson M (1994) Sexual selection. Princeton University Press, Princeton
Andersson M, Simmons LW (2006) Sexual selection and mate choice. Trends Ecol Evol 21:296–302. https://doi.org/10.1016/j.tree.2006.03.015
Barton K (2019) MuMIn: multi-model inference. R package version 1.43.6. https://CRAN.R-project.org/package=MuMIn
Bechsgaard J, Vanthournout B, Funch P, Vestbo S, Gibbs R, Richards S, Ichards K, Sanggaard K, Enghild J, Bilde T (2016) Comparative genomic study of arachnid immune systems indicates loss of beta-1,3-glucanase-related proteins and the immune deficiency pathway. J Evol Biol 29:277–291. https://doi.org/10.1111/jeb.12780
Bulet P, Stocklin R, Menin L (2004) Anti-microbial peptides: from invertebrates to vertebrates. Immunol Rev 98:169–184. https://doi.org/10.1111/j.0105-2896.2004.0124.x
Calbacho-Rosa L, Moreno-García MA, Lanz-Mendoza H, Peretti AV, Córdoba-Aguilar A (2012) Reproductive activities impair immunocompetence in Physocyclus dugesi (Araneae: Pholcidae). J Arachnol 40:18–22. https://doi.org/10.1636/Hill-13.1
Carballo M, Baldenegro F, Bollati F, Peretti AV, Aisenberg A (2017) No pain, no gain: male plasticity in burrow digging according to female rejection in a sand-dwelling wolf spider. Behav Process 140:174–180. https://doi.org/10.1016/j.beproc.2017.05.007
Contreras-Garduño J, Canales-Lazcano J, Córdoba-Aguilar A (2006) Wing pigmentation, immune ability, fat reserves and territorial status in males of the rubyspot damselfy, Hetaerina americana. J Ethol 24:165–173. https://doi.org/10.1007/s10164-005-0177-z
Córdoba-Aguilar A, Ruiz-Silva D, Munguia-Steyer R, Lanz-Mendoza H (2011) Do reproductive activities compromise immunological competence as measured by phenoloxidase activity? Field and experimental manipulation in females of two damselfly species. Physiol Entomol 36:335–342. https://doi.org/10.1111/j.1365-3032.2011.00802.x
Costa FG (1995) Ecología y actividad diaria de las arañas de la arena Allocosa spp. (Araneae, Lycosidae) en Marindia, localidad costera del Sur del Uruguay. Rev Bras Biol 55:457–466
Delignette-Muller ML, Dutang C (2015) R package for fitting distributions. J Stat Softw 64:1–34
Drury J (2010) Immunity and mate choice: a new outlook. Anim Behav 79:539–545. https://doi.org/10.1016/j.anbehav.2009.12.023
Dziarski R (2004) Peptidoglycan recognition proteins (PGRPs). Mol Immunol 40:877–886. https://doi.org/10.1016/j.molimm.2003.10.011
Fedorka KM, Mousseau TA (2006) Immune system activation affects male sexual signal and reproductive potential in crickets. Behav Ecol 18:231–235. https://doi.org/10.1093/beheco/arl067
Fedorka KM, Zuk M (2005) Sexual conflict and female immune suppression in the cricket, Allonemobius socius. J Evol Biol 18:1515–1522. https://doi.org/10.1111/j.1420-9101.2005.00942.x
Fedorka KM, Zuk M, Mousseau TA (2004) Immune suppression and the cost of reproduction in the ground cricket, Allonemobius socius. Evolution 58:2478–2485. https://doi.org/10.1111/j.0014-3820.2004.tb00877.x
Folstad I, Karter AJ (1992) Parasites, bright males, and the immunocompetence handicap. Am Nat 139:603–622. https://doi.org/10.1086/285346
Forsyth DM, Duncan RP, Tustin KG, Gaillard JM (2005) A substantial energetic cost to male reproduction in a sexually dimorphic ungulate. Ecology 86:2154–2163. https://doi.org/10.1890/03-0738Ç
Ghione S, Coelho L, Costa FG, García LF, González M, Jorge C, Laborda A, Montes de Oca L, Pérez-Miles F, Postiglioni R, Simó M, Toscano-Gadea C, Viera C, Aisenberg A (2017) Arácnidos prioritarios para la conservación en Uruguay. Bol Soc Zool Uruguay 26:1–8
Gilbert R, Karp RD, Uetz GW (2016) Effects of juvenile infection on adult immunity and secondary sexual characters in a wolf spider. BehavEcol 27:946–954. https://doi.org/10.1093/beheco/arv241
Gillespie JP, Kanost MR, Trenczek T (1997) Biological mediators of insect immunity. Annu Rev Entomol 42:611–643. https://doi.org/10.1146/annurev.ento.42.1.611
Gwynne DT, Watkiss J (1975) Burrow blocking behaviour in Geolycosa wrightii (Araneae: Lycosidae). Anim Behav 23:953–956. https://doi.org/10.1016/0003-3472(75)90120-7
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387. https://doi.org/10.1126/science.7123238
Henschel JR (1990) The biology of Leucorchestris arenicola (Araneae: Heteropodidae), a burrowing spider of the Namib dunes. In: Seely MK (ed) Namib ecology: 25 years of Namib research. Transvaal Museum Monograph No. 7, Pretoria, pp 115–127
Imler JL (2014) Overview of Drosophila immunity: a historical perspective. Dev Comp Immunol 42:3–15. https://doi.org/10.1016/j.dci.2013.08.018
Jacot A, Scheuber H, Kurtz J, Brinkhof MW (2005) Juvenile immune status affects the expression of a sexually selected trait in field crickets. J Evol Biol 18:1060–1068. https://doi.org/10.1111/j.1420-9101.2005.00899.x
Janeway CA, Medzhitov R (2002) Innate immune recognition. Annu Rev Immunol 20:197–216. https://doi.org/10.1146/annurevlinmunol.20.083001.084359
Johnstone RA (1995) Honest advertisement of multiple qualities using multiple signals. J Theoret Biol 177:87–94. https://doi.org/10.1016/S0022-5193(05)80006-2
Kim YS, Ryu JH, Hani SJ, Choi KH, Nam KB, Jang IH, Lemaitre B, Breyi PT, Lee WJ (2000) Gram-negative bacteria-binding protein, a pattern recognition receptor for lipopolysaccharide and beta1,3-glucan that mediates the signaling for the induction of innate immune genes in drosophila melanogaster cells. J Biol Chem 275:32721–32727. https://doi.org/10.1074/jbc.M003934200
Kotiaho JS (2000) Testing the assumptions of conditional handicap theory: costs and condition dependence of a sexually selected trait. Behav Ecol Sociobiol 48:188–194. https://doi.org/10.1007/s002650000221
Kuhn-Nentwig L, Nentwig W (2013) The immune system of spiders. In: Nentwig W (ed) Spider ecophysiology. Springer, Berlin, pp 81–91
Lawniczak M, Barnes A, Linklater J, James M, Boone S, Tracey C (2006) Mating and immunity in invertebrates. Trends Ecol Evol 22:48–55. https://doi.org/10.1016/j.tree.2006.09.012
Lemaitre B, Hoffmann J (2007) The host defense of Drosophila melanogaster. Annu Rev Immunol 25:697–743. https://doi.org/10.1016/j.tree.2006.09.012
Loof AD (2011) Longevity and aging in insects: is reproduction costly; cheap; beneficial or irrelevant? A critical evaluation of the ‘trade-off’ concept. J Insect Physiol 57:1–11. https://doi.org/10.1016/j.jinsphys.2010.08.018
Mappes J, Alatalo RV, Kotiaho J, Parri S (1996) Viability costs of condition-dependent sexual male display in a drumming wolf spider. Proc R Soc Lond B 263:785–789. https://doi.org/10.1098/rspb.1996.0117
Møller P, Saino N (1994) Parasites, Immunology of hosts, and host sexual selection. J Parasitol 80:850–858. https://doi.org/10.2307/3283433
Møller AP, Christe P, Erritzoee J, Mavarez J (1998) Condition, disease and immune defence. Oikos 83:301–306 https://www.jstor.org/stable/3546841
Møller AP, Christe P, Lux E (1999) Parasitism, host immune function, and sexual selection. Q Rev Biol 74:3–20. https://doi.org/10.1086/392949
Monroy KJ, Korb J (2016) Ageing and the reversal of the fecundity/longevity trade-off in social insects. Curr Opin Insect Sci 16:7–10. https://doi.org/10.1371/journal.pone.0210371
Moya-Laraño J, Pascual J, Wise DH (2003) Mating patterns in late-maturing female Mediterranean tarantulas may reflect the costs and benefits of sexual cannibalism. Anim Behav 66:469–476. https://doi.org/10.1006/anbe.2003.2262
Muta T, Iwanaga S (1996) The role of hemolympyh coagulation in innate immunity. Curr Opin Immunol 8:41–47. https://doi.org/10.1016/S0952-7915(96)80103-8
Oku K, Price T, Wedell N (2019) Does mating negatively affect female immune defences in insects? Anim Biol 69:117–136. https://doi.org/10.1163/15707563-20191082
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Raberg L, Grahn M, Hasselquist D, Svensson E (1998) On the adaptive significance of stress-induced immunosuppression. Proc R Soc Lond B 265:637–1641. https://doi.org/10.1098/rspb.1998.0482
Ragan EJ, An C, Jiang H, Kanost MR (2009) Roles of haemolymph proteins in antimicrobial defences of Manduca sexta. In: Insect Infection and Immunity. Oxford University Press, pp 34–48
Rantala MJ, Koskimäki J, Taskinen J, Tynkkynen K, Suhonen J (2000) Immunocompetence, developmental stability and wingspot size in the damselfly Calopteryx splendens L. Proc R Soc Lond B 267:2453–2457. https://doi.org/10.1098/rspb.2000.1305
Rantala MJ, Jokinen I, Kortet R, Vainikka A, Suhonen J (2002) Do pheromones reveal male immunocompetence? Proc R Soc Lond B 269:1681–1685. https://doi.org/10.1098/rspb.2002.2056
Rigby MC, Jokela J (2000) Predator avoidance and immune defense: costs and trade-offs in snails. Proc Biol Sci 267:171–176. https://doi.org/10.1098/rspb.2000.0983
Roberts ML, Buchanan KL, Evans MR (2004) Testing the immunocompetence handicap hypothesis: a review of the evidence. Anim Behav 68:227–239. https://doi.org/10.1016/j.anbehav.2004.05.001
Rolff J, Siva-Jothy MT (2002) Copulation corrupts immunity: a mechanism for a cost of mating in insects. Proc Nat Acad Sci 99:9916–9918. https://doi.org/10.1073/pnas.152271999
Rowley AF, Powell A (2007) Invertebrate immune systems–specific, quasi-specific, or nonspecific? J Immunol 179:7209–7214. https://doi.org/10.4049/jimmunol.179.11.7209
Ruiz-Guzmán G, Canales-Lazcano J, Jiménez-Cortés J, Contreras-Garduño J (2013) Sexual dimorphism in immune response: testing the hypothesis in an insect species with two male morphs. Insect Sci 20:620–628. https://doi.org/10.1111/j.1744-7917.2012.01551.x
Ryder JJ, Siva-Jothy MT (2000) Male calling song provides a reliable signal of immune function in a cricket. Proc R Soc Lond B 267:1171–1175. https://doi.org/10.1098/rspb.2000.1125
Sapolsky RM (1992) Neuroendocrinology of the stress response. In: Becker JB, Breedlove SM, Crews D (eds) Behavioural endocrinology. MIT Press, Cambridge, pp 287–324
Schmid-Hempel P (2003) Variation in immune defence as a question of evolutionary ecology. Proc R Soc B 270:357–366. https://doi.org/10.1098/rspb.2002.2265
Schmid-Hempel P (2005) Evolutionary ecology of insect immune defenses. Annu Rev Entomol 50:529–551. 10.1146.annurev.ento.50.071803.130420
Seitz KA (1972) Zur Histologie und Feinstruktur des Herzens und der Hamocyten von Cupiennius salei KEYS (Araneae, Ctenidae). Zool J Anat 89:351–384
Sheldon BC, Verhulst S (1996) Ecological immunology: costly parasite defenses and trade-offs in evolutionary ecology. Trends Ecol Evol 11:317–321. https://doi.org/10.1016/0169-5347(96)10039-2
Shutler D (2010) Sexual selection: when to expect trade-offs. Biol Lett 7:101–104. https://doi.org/10.1098/rsbl.2010.0531
Simmons LW (2011) Resource allocation trade-off between sperm quality and immunity in the field cricket, Teleogryllus oceanicus. Behav Ecol 23:168–173. https://doi.org/10.1093/beheco/arr170
Simó M, Lise A, Pompozzi G, Laborda Á (2017) On the taxonomy of southern South American species of the wolf spider genus Allocosa (Araneae: Lycosidae: Allocosinae). Zootaxa 4216:261–278. https://doi.org/10.11646/zootaxa.4216.3.4
Siva-Jothy MT, Thompson JJ (2002) Short-term nutrient deprivation affects immune function. Physiol Entomol 27:206–212. https://doi.org/10.1046/j.1365-2.2002.00286.x
Siva-Jothy MT, Tsubaki Y, Hooper RE (1998) Decreased immune response as a proximate cost of copulation and oviposition in a damselfly. Physiol Entomol 23:274–277. https://doi.org/10.1046/j.1365-3032.1998.233090.x
Venables WN, Ripley BD (2002) Exploratory multivariate analysis. In: Modern applied statistics with S. Springer, New York, pp 301–330
Zahavi A (1975) Mate selection a selection for a handicap. J Theor Biol 53:205–214
Zuk M (1990) Reproductive strategies and disease susceptibility: an evolutionary viewpoint. Parasitol Today 6:231–233. https://doi.org/10.1016/0169-4758(90)90202-F
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. I J Parasitol 26:1009–1024. https://doi.org/10.1016/S0020-7519(96)80001-4
Zuk M, Stoehr AM (2002) Immune defense and host life history. Am Nat 160:S9–S22. https://doi.org/10.1086/342131
Acknowledgments
We thank M. González, R. Postiglioni, L. Bidegaray, M. Carballo, and E. Stanley for the help during fieldwork. We also thank M. Laviña (Sección Microbiología) for providing the lysozyme. We are grateful to A. Díaz, G. González, M. Rosotti, and M. Pires for their support in providing the equipment and for their help during lab work. A. Dipaolo and Dr. Miguel Moreno collaborated with the technique. The authors want to thank Instituto de Higiene, Cátedra de Inmunología, Facultad de Química, Montevideo, Uruguay, for providing the laboratory for developing the lytic activity analyses. L.C.R. and F.C. would also like to thank the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET-Argentina). We thank the Editor, Matjaž Gregorič, Simona Kralj-Fišer, and anonymous referees who provided helpful and constructive comments that improved the final version of the manuscript. Finally, we are grateful to Ted Henderson for his grammar revision of the text.
Funding
A. Albín was financially supported by the Agencia Nacional de Investigación e Innovación under the code POS_NAC_2013_1_555. A. Aisenberg and M. Simó acknowledge financial support by PEDECIBA (Programa de Desarrollo de Ciencias Básicas), and SNI (ANII). L. Calbacho-Rosa acknowledges financial support by CONICET (Consejo Nacional de Ciencia y Tecnología — Argentina).
Author information
Authors and Affiliations
Contributions
All the authors designed the experiments and A. Albín (A.R.A.) and L.C. performed them. A.R.A., A.A., and M.S. collected the spiders. A.R.A., L.C., and F.C. performed the data analysis. All the authors contributed with bibliography and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Additional information
Communicated by: Matjaž Gregorič
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Albín, A., Simó, M., Cargnelutti, F. et al. Sex and burrowing behavior and their implications with lytic activity in the sand-dwelling spider Allocosa senex. Sci Nat 107, 44 (2020). https://doi.org/10.1007/s00114-020-01700-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-020-01700-2